-
Is Matter Around Us Pure
*Saturated solution :–
A solution which at a given temperature dissolves as much solute as it is capable of dissolving, is said to be a saturated solution.
Ex.At 30°C, 55 g of common salt dissolves in 100g of water. However, if more of common salt is added to the above solution, it just does not dissolve. In such a situation,the solution of common
salt containing 55 gm of salt in 100 gm of water, is a saturated solution at 30°C.
If a saturated solution at some particular temperature is heated, the solution becomes unsaturated, because of the increase in solubility.
If a saturated solution at some higher temperature is cooled, it remains saturated. The excess
solute comes out of the solution and deposits it self in the form of crystals.
*Unsaturated solution :–
When the amount of solute contained in a solution is less than the saturation level, the solution is said to be an unsaturated solution.
Ex.At 30°C, if 45 g of common salt is dissolved in 100 g of water, such solution so formed is capable of dissolving more of the common salt, then such a solution is called unsaturated solution.
*Super saturated solution :–
A solution which contains more of the solute than required to make a saturated solution, is called a super saturated solution.
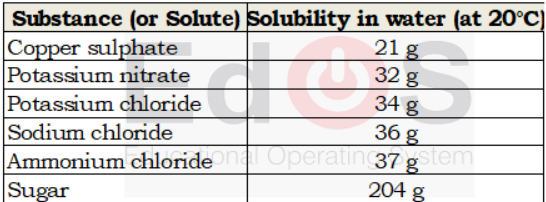
*Solubility of a solute :– The amount of solute (in gram), which dissolve in 100 g of water (solvent), at a given temperature, is called solubility of the solute at that temperature.