-
Thrust And Pressure
BAROMETER
The instrument used for measuring of pressure is called Barometer. It was constructed by Torricelli in 1643.
It consists of a glass tube with a narrow uniform bore and of length 100 cm. It is closed at one end. The tube is carefully filled with perfectly pure and dry mercury so that no air bubbles are left inside the tube.
Thumb is placed tightly on the open end of the tube and then it is inverted in a bowl containing mercury.
The thumb is removed from the open end, under the surface of mercury in the bowl. It is observed that some mercury flows down, thereby creating an empty space which is commonly called Torricellian vacuum.
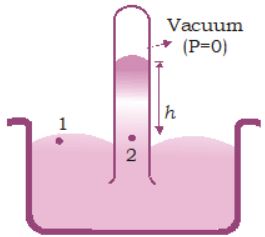
In principle, any liquid can be used to fill the barometer, but mercury is the substance of choice because its great density makes possible an instrument of reasonable size.
P1 = P2
Here, P1 = atmospheric pressure (P0) and \( P_2 = 0 + \rho gh = \rho gh \)
Here, \(\rho\) density of mercury
\( \therefore P_0 = \rho gh \)
Thus, the mercury barometer reads the atmospheric pressure (P0) directly from the height of the mercury column. For example if the height of mercury in a barometer is 760 mm, then atmospheric pressure will be,
\( P_0 = \rho gh \) = (13.6 × 103)(9.8)(0.760) = 1.01 × 105 N/m2