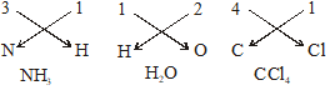-
Ions And Formula
Writing formula of a Binary Molecular Compound
In binary molecular compounds, the atoms of the two elements are held by covalent bonds. A covalent bond, as you know, is formed by sharing of electrons between the two atoms. The number of electrons in an atom of the element contributes for sharing is known as valency of the element. Knowing the valencies of the two elements involved in the formation of a binary molecular compound, the formula of the compound can be derived.
Step.1 Write the constituent elements and their valencies as shown below :
Valency 4 2
Element C O
Step.2 Reduce the valency numerals to simplest whole numbers by dividing by some common factor, if any.
2 1
C O
Step.3 Criss cross the reduced valency numerals and write them as subscripts at
bottom right hand side of the symbols.
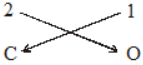
The subscript 1 is not written. Thus, the formula of the compound is CO2.
Formulae of some more compounds are given below :