-
Interconversion Of Matter
EFFECT OF CHANGE OF TEMPERATURE
Effect of temperature on the change of state of matter can be explained by the following experiment
*Increasing the temperature by heating
Experiment: Take about 150g of ice in a beaker and suspend a laboratory thermometer so that its bulb is in contact with the ice (figure 5). Now start heating the beaker.
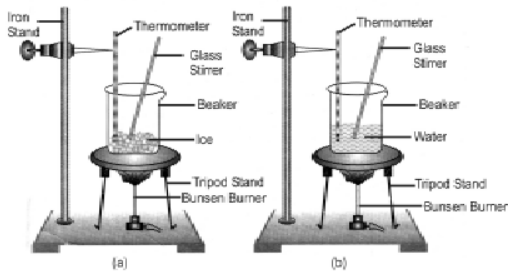
Observation: On heating, it will be observed that first the ice (solid) melts to form water (liquid). If the heating is carried out further, the liquid (water) will change to gaseous state (vapour)
Discussion :-
(i) Change of state from solid to liquid (fusion): When heat is supplied to a solid (ice), the kinetic energy of solid particles increases due to increase in temperature. As a result, solid particles start vibrating with high speed. On further increasing temperature, the heat energy overcomes the forces of attraction between solid particles. At this temperature, the particles leave their fixed positions, start flowing and thus solid melts to form a liquid.
The temperature at which a solid melts to become a liquid at atmospheric pressure is called its ‘melting point’. This process of change of solid state into liquid state is also called ‘Fusion’.
\( Solid \to Liquid \)The melting point of ice is O0C or 273 K (O0C = 273 + 0 = 273K). This temperature (i.e. O0C) remains constant till all the ice has melted even though we continue to supply the heat. This is because, the heat energy supplied is absorbed by the particles of ice to overcome the forces of attraction between them to change them from solid to liquid state without showing any rise in temperature. Therefore, it is considered that this heat gets hidden within the particles and is thus called latent heat (latent means hidden).
(ii) Change of state from liquid to gas (vaporisation) : When heat is further supplied to the liquid, kinetic energy of liquid particles increase further, as a result of this, inter-particle distance increase (app. 100 times). Hence, the magnitude of forces of attraction holding the liquid particles becomes so less that, the liquid particles break apart from the liquid state and change to gaseous or vapour state. The pressure of air in atmosphere is called atmospheric pressure.
\( Liquid \to Gas \)
Each pure liquid has a fixed boiling point. The boiling point of pure water is 1000C or 373 K (1000C = 273 + 100 = 373K) This temperature (i.e. 1000C) again becomes constant till all the liquid has vaporized. This is again because that heat energy supplied is absorbed by the liquid water particles to overcome the forces of attraction between them to change from liquid water to steam, without showing any rise in temperature. In other words, heat gets hidden within the particles and is therefore, called latent heat.