-
Acids, Bases & Salts - Indicators
INDICATORS
For identifying a substance as an acid or a base we make use of certain natural chemical substances which show a change in colour when placed in acid medium, base medium. These chemical substances are called indicators.
Turmeric is yellow in colour and stains the clothing yellow. Rubbing with soap or detergent, its colour charged to brownish red because soap solution contains a base. Again by rubbing with lemon piece stain regains its yellow colour because lemon contains acid in it.
So, turmeric is an indicator
Turmeric is another natural indicator
Activity
Ø Take a tablespoonful of turmeric powder. Add a little water and make a paste.
Ø Make turmeric paper by depositing turmeric paste on blotting paper/filter paper and rying it. Cut thin strips of the yellow paper obtained.
Ø Put a drop of soap solution on the strip of turmeric paper.
What do you observe?
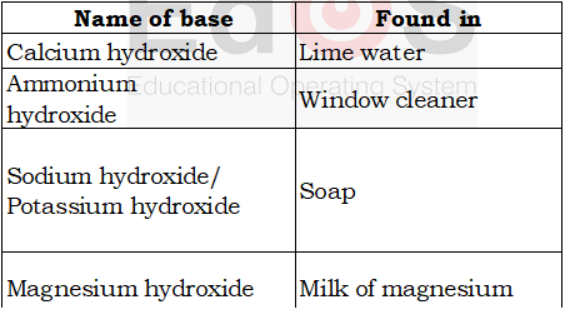
To prepare limewater, dissolve some lime (chuna) in water in a bottle. Stir the solution and keep it for some time. Pour a little from the top. This is lime water.

A substance acts as an indicator which has a different colour in acid and different colour in base solution.
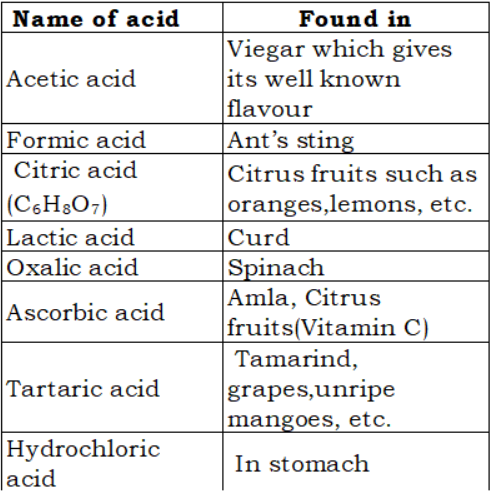
All the acids mentioned above occur in nature
Litmus:
Litmus extracted from naturally occurring plants, the lichens. Litmus change its colour to red in an acidic solution and it changes its colour to blue in basic or alkaline solution. Litmus paper is prepared by soaking rectangular strips of filler paper in litmus solution and then drying them.
Methyl orange:
Methyl orange solution is pale yellow in colour. It turns pink in acidic solution.
Phenolphthalein:
Its solution is prepared in alcohol. Its solution remains colourless in acidic solution. It turns pink in basic or alkaline solution.