-
Ions And Formula
Writing formula of An Ionic Compound (or) Compound containing Radicals:
The formula of an ionic compound represents the simplest whole number ratio of ions in it. The total positive charge on cations is equal to the total negative charge on anions. Knowing the formulae of the radicals present in the compound, the formula of the compound can be written by the following steps :
Step.1 Write the formulae of the radicals of the compound side by side with basic radical (Cation or Electropositive ion) on the left hand side and acid radical (Anion or Electronegative ion ) on the right hand side.
Step.2 Enclose the compound radical (if any) in a bracket.
Step.3 Reduce the valency numerals to a simple ratio by dividing with a common factor, if any.
Step.4 Criss-cross the valencies, i.e., shift the valency numerals crosswise to the lower right hand corner of the radicals. The is done to achieve electrical neutrality.
Let us apply the above steps to write formula of calcium phosphate.
Step.1 Writing the formulae of the radicals.
Ca2+ PO43–
Step.2 Enclose the compound radical phosphate in bracket.
Ca2+ [PO4]3–
Step.3 Not applicable, because ratio is already simple.
Step.4 Criss-cross the valencies.

Thus, the formula of calcium phosphate is : Ca3(PO4)2
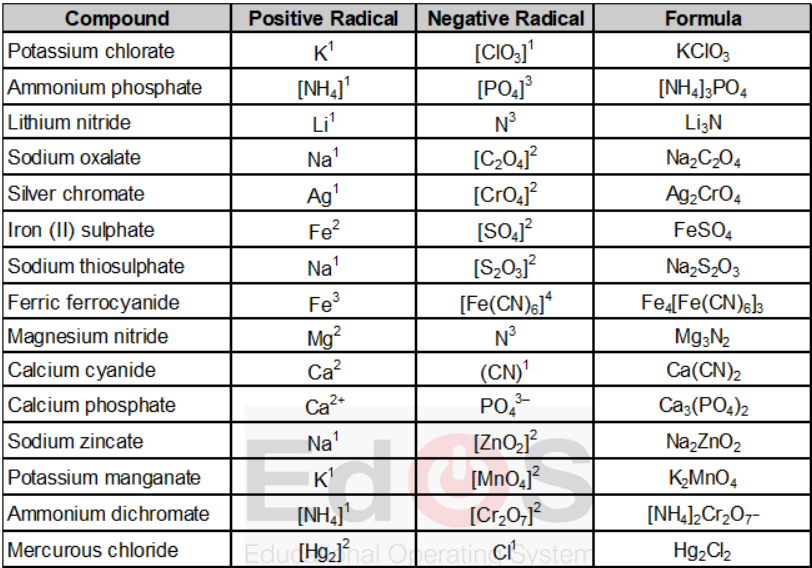
On the basis of above steps the formula of some substances are given below in table