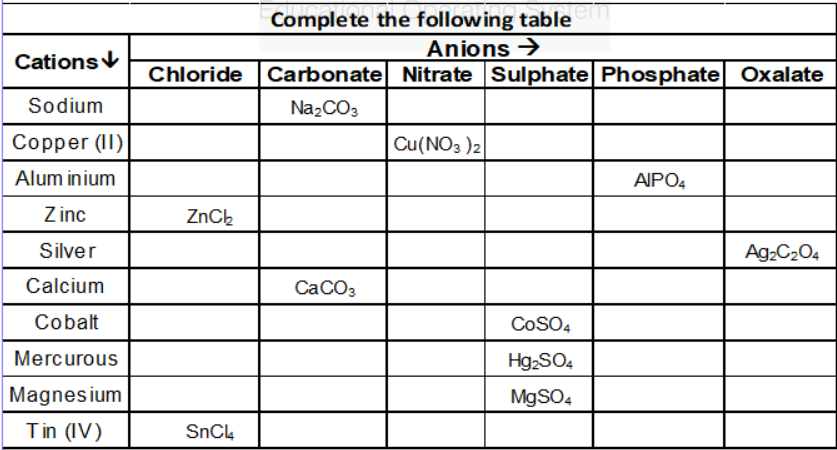
-
Ions And Formula
Worked Examples
Example-1
Write the Formula of oxide of elements ‘X’ when its Valency is 3
Solution
Symbols : X O
Valencies : 3 2
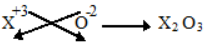 (Formula)
(Formula)
Example-2
If an Element ‘E’ has valency of 2.
i) What will be the formulae of it’s hydroxide ?
ii) What will be the formulae of its sulphide ?
Solution
i) Formulae of hydroxide of elements ‘E’
Symbols : E 0H
Valencies : 2 1
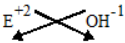
Cross-over valencies.
E1 OH2 (or) E (OH)2
Formulae of hydroxide is E (OH)2
ii) Formulae of Sulphide of element ‘E’
Symbols : E S
Valencies : 2 2

Cross-over valencies.
E2 S2 (or) ES
Formulae of Sulphide is ES
Practice Corner