-
Introduction To Chemical Change
CHEMICAL CHANGE
A change with which you are quite familiar is the rusting of iron. If you leave a piece of iron in the open for some time, it acquires a film of brownish substance. This substance is called rust and the process is called rusting (Fig. 6.2). Iron gates of parks or farmlands, iron benches kept in lawns and gardens, almost every article of iron, kept in the open gets rusted. At home you must have seen shovels and spades getting rusted when exposed to the
Fig. 6.2 Rusting iron
atmosphere for some time. In the kitchen, a wet iron pan (tawa) often gets rusted if left in that state for some time. Rust is not iron. It is different from iron on which it gets deposited.
Let us consider a few more changes where new substances are formed.
-
Reaction With Magnesium
Activity 6.6
(To be demonstrated by the teacher)
Get a small piece of a thin strip or ribbon of magnesium. Clean its tip with sandpaper. Bring the tip near a candle flame. It burns with a brilliant white light (Fig. 6.3). When it is completely burnt it leaves behind a powdery ash.
Fig. 6.3 Magnesium ribbon burning
Does the ash look like the magnesium ribbon?
The change can be represented by the following equation:
Magnesium (Mg) + Oxygen (O2 )------>Magnesium oxide (MgO)
The equations here are different from those in mathematics. In equations of this kind, the arrow implies ‘becomes’. No attempt should be made to balance chemical equations at this stage.
Collect the ash and mix it with a small amount of water. Stir the mixture (aqueous solution) well. Test the mixture with blue and red litmus papers.
Does the mixture turn red litmus blue?
Does the mixture turn blue litmus red?
On the basis of this test, how do you classify the aqueous solution — acidic or basic?
On dissolving the ash in water it forms a new substance. This change can be written in the form of the following equation:
Magnesium oxide + Water → Magnesium hydroxide
As you have already learnt in Chapter 5, magnesium hydroxide is a base. So, magnesium oxide is a new substance formed on burning of magnesium. Magnesium hydroxide is another new substance formed by mixing magnesium oxide with water.
-
Reaction Between Iron And Copper Sulphate
Activity 6.7
(To be demonstrated by the teacher) Dissolve about a teaspoonful of copper sulphate (blue vitriol or neela thotha) in about half a cup of water in a glass tumbler or a beaker. Add a few drops of dilute sulphuric acid to the solution. You should get a blue coloured solution. Save a small sample of the solution in a test tube or a small glass bottle. Drop a nail or a used shaving blade into the remaining solution. Wait for half an hour or so. Observe the colour of the solution. Compare it with the colour of the sample solution saved separately (Fig. 6.4).
Fig. 6.4 Change in colour of the copper sulphate solution due to reaction with iron
Do you see any change in the colour of the solution?
Take out the nail or the blade. Has it changed in any way?
The changes that you notice are due to a reaction between copper sulphate and iron. The change of
colour of the solution from blue to green is due to the formation of iron sulphate, a new substance. The brown deposit on the iron nail is copper, another new substance. We can write the reaction as:
Copper sulphate solution (blue) + Iron → Iron sulphate solution (green)+ Copper (brown deposit)
-
Reaction Between Iron And Copper Sulphate
Activity 6.7
(To be demonstrated by the teacher) Dissolve about a teaspoonful of copper sulphate (blue vitriol or neela thotha) in about half a cup of water in a glass tumbler or a beaker. Add a few drops of dilute sulphuric acid to the solution. You should get a blue coloured solution. Save a small sample of the solution in a test tube or a small glass bottle. Drop a nail or a used shaving blade into the remaining solution. Wait for half an hour or so. Observe the colour of the solution. Compare it with the colour of the sample solution saved separately (Fig. 6.4).
Fig. 6.4 Change in colour of the copper sulphate solution due to reaction with iron
Do you see any change in the colour of the solution?
Take out the nail or the blade. Has it changed in any way?
The changes that you notice are due to a reaction between copper sulphate and iron. The change of
colour of the solution from blue to green is due to the formation of iron sulphate, a new substance. The brown deposit on the iron nail is copper, another new substance. We can write the reaction as:
Copper sulphate solution (blue) + Iron → Iron sulphate solution (green)+ Copper (brown deposit)
-
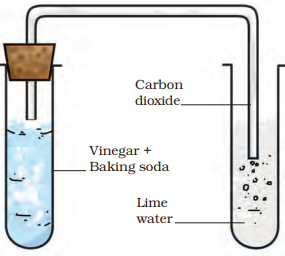
Reaction Of Vinegar With Baking Soda
Activity 6.8
Take about a teaspoonful of vinegar in a test tube. Add a pinch of baking soda to it. You would hear a hissing sound and see bubbles of a gas coming out. Pass this gas through freshly prepared lime water as shown in Fig. 6.5.
What happens to the lime water?
The change in the test tube is as follows:
Vinegar (Acetic acid) + Baking soda (Sodium hydrogencarbonate)→ Carbon dioxide + other substances The reaction between carbon dioxide and lime water is as follows:
Fig. 6.5 Set up to pass gas through lime water
Carbon dioxide (CO2) + Lime water [Ca(OH)2] →Calcium Carbonate (CaCO3) + Water (H2O)
When carbon dioxide is passed through lime water, calcium carbonate is formed, which makes lime water milky. The turning of lime water into milky is a standard test of carbon dioxide. You will use it in Chapter 10 to show that the air we breathe out is rich in carbon dioxide.
In Activities 6.6–6.8, you saw that in each change one or more new substances were formed. In Activity 6.6, the ash was the new substance formed when magnesium was burnt in air. In Activity 6.7, the reaction of copper sulphate with iron produced iron sulphate and copper. Both of these are new substances. Copper was deposited on the shaving blade of iron. In Activity 6.8, vinegar and baking soda together produced carbon dioxide, which turned lime water milky. Can you name the new substance formed in this reaction?
-
Rusting Of Iron
RUSTING OF IRON
Let us get back to rusting. This is one change that affects iron articles and slowly destroys them. Since iron is
used in making bridges, ships, cars, truck bodies and many other articles, the monetary loss due to rusting is huge.
The process of rusting can be represented by the following equation:
Iron (Fe) + Oxygen (O2 , from the air) + water (H2O) → rust (iron oxide Fe2O3 )
For rusting, the presence of both oxygen and water (or water vapour) is essential.
In fact, if the content of moisture in air is high, which means if it is more humid, rusting becomes faster.
So, how do we prevent rusting? Prevent iron articles from coming in contact with oxygen, or water, or both.
One simple way is to apply a coat of paint or grease. In fact, these coats should be applied regularly to prevent rusting. Another way is to deposit a layer of a metal like chromium or zinc on iron.
This process of depositing a layer of zinc on iron is called galvanisation. The iron pipes we use in our homes to carry water are galvanised to prevent rusting.
You know that ships are made of iron and a part of them remains under water. On the part above water also, water drops keep clinging to the ship’s outer surface. Moreover, the water of the sea contains many salts. The salt water makes the process of rust formation faster. Therefore, ships suffer a lot of damage from rusting in spite of being painted. So much so, that a fraction of ship’s iron has to be replaced every year. Can you imagine the monetary loss to the world?
Stainless steel is made by mixing iron with carbon and metals like chromium, nickel and manganese. It does not rust.
-
Chemical Reaction
A change in which one or more new substances are formed is called a chemical change. A chemical change is also called a chemical reaction.
Chemical changes are very important in our lives. All new substances are formed as a result of chemical changes. For example, digestion of food in our body, ripening of fruits, fermentation of grapes, etc., happen due to series of chemical changes. A medicine is the end product of a chain of chemical reactions. Useful new materials, such as plastics and detergents, are produced by chemical reactions. Indeed, every new material is discovered by studying chemical changes.
We have seen that one or more new substances are produced in a chemical change. In addition to new products, the following may accompany a chemical change:
1.Heat, light or any other radiation (ultraviolet, for example) may be given off or absorbed.
2.Sound may be produced.
3.A change in smell may take place or a new smell may be given off.
4.A colour change may take place .
5.A gas may be formed.
Let us look at some examples
You saw that burning of magnesium ribbon is a chemical change. Burning of coal, wood or leaves is also a chemical change. In fact, burning of any substance is a chemical change. Burning is always accompanied by production of heat.
Explosion of a firework is a chemical change. You know that such an explosion produces heat, light, sound and unpleasant gases that pollute the atmosphere. That is why you are advised not to play with fireworks.
When food gets spoiled, it produces a foul smell. Shall we call this change a chemical change?
You must have noticed that a slice of an apple acquires a brown colour if it is not consumed immediately. If you have not seen this change in colour, cut a fresh slice of apple and keep it away for some time. Repeat the same activity with a slice of potato or brinjal. The change of colour in these cases is due to the formation of new substances. Are not these changes chemical changes?
In Chapter 5, you neutralised an acid with a base. Is neutralisation a chemical change?
-
Crystallization
CRYSTALLIZATION
In Class VI you have learnt that salt can be obtained by the evaporation of sea water. The salt obtained in this manner is not pure and the shape of its crystals cannot be seen clearly. However, large crystals of pure substances can be formed from their solutions. The process is called crystallisation. It is an example of a physical change.
Activity 6.9
(To be performed in the presence of the teacher)
Take a cupful of water in a beaker and add a few drops of dilute sulphuric acid. Heat the water. When it starts boiling add copper sulphate powder slowly while stirring continuously (Fig. 6.6). Continue adding copper sulphate powder till no more powder can be dissolved. Filter the solution. Allow it to cool. Do not disturb the solution when it is cooling. Look at the solution after some time. Can you see the crystals of copper sulphate? If not, wait for some more time.
Fig. 6.6 Crystals of copper sulphate
You have learnt about physical and chemical changes.Try to identify changes that you observe around you as physical or chemical changes.