-
NATURE OF MATTER -CHARACTERISTICS OF MATTER
MATTER: Any material which has mass and occupies space is called matter.
CHARACTERISTICS OF MATTER :
(i) Matter is composed of tiny particles called molecules, inturn made up of atoms.
(ii) All material bodies have mass.
(iii) All material bodies occupy space.
We can say that all the object around us are composed of matter.e.g. chair, pencil , air, oil etc.
These particles have intermolecular spaces between them and attract each other with a force and are in continuos motion.
Matters are classified in two ways :
(i) On the basis of physical states, all the matter can be classified into three groups: Solids, Liquids and Gases.
(ii) On the basis of chemical constitution as an element, compound or mixture.
Matter exists in five states :
1.Solids 2. Liquids 3.Gases 4. Plasma 5.Bose-Einstein condensate.
SOLIDS:
In solids, the molecules are closely packed, when heated at melting point, it changes its physical state and becomes a liquid. For an instance, When a solid substance is heated, the heat energy make its molecules vibrate more vigorously. At the melting point, the molecules of a solid have sufficient kinetic energy to overcome the strong forces of attraction holding them in fixed positions and break to form small groups of molecules.Hence the solid melts to form a liquid.
1)A solid has fixed shape
2) A solid has fixed size
3) A solid can not be compressed
4) A solid does not fill its container completely.
5) A solid does not flow.
Examples: Wood,Stone,Sand,Iron,Ice etc
Liquids
The spaces between the molecules of a liquid are slightly more than that in a solid but they are still very small. Due to comparatively less strong intermolecular forces, the positions of molecules in a liquid are not fixed. The molecules of a liquid can move from one position to another within the liquid.
The molecules of a liquid have more kinetic energy than the molecules of a solid.
1. A liquid does not have a fixed shape
2. A liquid has a fixed volume
3. A liquid cannot be compressed much
4. A liquid has comparatively high density
5. A liquid does not fill its container completely
6. A liquid generally flows easily. Ex: Milk, Water etc.
GASES:
The molecules of a gas have the maximum movement (or maximum kinetic energy). Due to this, the gases have the most disorder arrangement of molecules. Because of high kinetic energy and negligible forces of attraction, the molecules of a gas move with high speed in all directions. When the fast moving gas molecules hit the walls of the container, they exert a pressure (called gas pressure). Thus, a gas exerts a pressure on the walls of its container. The pressure exerted by a gas is due to the collisions of the fast moving gas molecules against the walls of the container. When a gas is put in an empty container, it quickly spreads
throughout the container and fills it completely.
1. A gas does not have a fixed shape 2. A gas does not have a fixed volume
3. A gas can be compressed easily 4. A gas has very low density
5. A gas fills its container completely 6. A gas flows easily.
Ex:Air, Oxygen etc....
PLASMA :
Plasma is the fourth state of matter.
Plasma is an ionised gas, a gas to which sufficient energy is provided to get free electrons from atoms or molecules to allow both the species, ions and electrons to co-exist. It exists in the presence of high temperature
Ex: The gases in the sun and stars exist in plasma state.
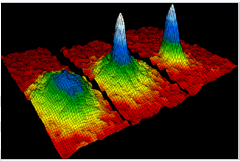
BOSE-EINSTEIN CONDENSATE:
It is the fifth state of matter.
Bose-Einstein condensate is a super fluid phase formed by atoms, cooled very near to absolute zero temperature. It is opposite to plasma state as it occurs at very low temperature.
Ex: First condensate was produced by Rubidium atoms (Rb) cooling to 170 nanokelvin.