-
Kinetic molecular theory of matter
PHYSICAL AND CHEMICAL CLASSIFICATION OF MATTER
Matter is defined as something which has some mass and occupies some space. It occurs in three different states based on physical classification. They are solids, liquids & gases. These three states of matter differ in intermolecular forces.
Kinetic molecular theory of matter :
This theory can be explained in a few postulates as follows.
1. Matter consists of a large number of extremely small particles called molecules.
2. In gaseous state the molecules can freely move, but in solid state the molecules
are not free to move.
3. The energy possessed by molecules as a result of motion is called kinetic energy.
4. Kinetic energy of gas molecules is directly proportional to absolute temperature. If temperature is more kinetic energy is more. If the temperature is less kinetic energy is less.
5. In gaseous state the molecules move completely independent of one another.
6. The pressure exerted by gas molecules is due to the bombardment of moving molecules on the walls of the container. -
Properties of different states of matter
Properties of different states of matter based on physical classification:
The three states of matter exhibit different properties. Their properties can be shown in the form of a table as follows.
Interconversion of states of matter :
The method of changing the matter from one state to another state by altering the conditions of temperature and pressure & back to the original state is called interconversion of state.
Conversion of solid to liquid & Vice versa:
The method of converting solid to liquid is called fusion or melting. The reverse process i.e. Conversion of liquid to solid state is called solidification or freezing. For melting,energy should be provided and for freezing energy should be taken away.
Melting point :
The temperature at which solid & liquid states co-exist is called melting point. The purity of solid substance can be decided based on melting point. A pure substance will have sharp and definite melting point. The melting point of ice is 0OC.
Freezing :
The process of conversion of liquid to solid by giving out heat eneryg is called freezing or solidification. When heat is extracted kinetic energy decreases and temperature also decreases. It is the reverse process of melting.
The constant temperature at which liquid changes to solid at atmospheric pressure is called freezing point. The freezing point of water is 0OC.
Conversion of liquid into gaseous state :
It is a known fact that liquids can be converted into gases by heating and gases can be converted into liquids by cooling. The process of converting a liquid to a gas is called Vaporisation. When the liquid is heated to a very high temperature, kinetic energy increases and the liquid boils off. The temperature as which a liquid boils under an external pressure of one atmosphere is called normal boiling point of the liquid.
Condensation or Vaporisation :
The process of converting a gas or Vapour into liquid is called condensation. When heat energy is extracted from vapour molecules, kinetic energy decreases, as a result of it temperature decreases. The condensation point or boiling point is 100 OC Conversion of a gas to liquid can be carried out by the application of external pressure. But for every gas there a certain temperature called critical temperature. Above this temperature the gas can not be converted into liquid even if high pressure is applied.
Conversion of solid into gas and gas to solid :
The method of conversion of a solid into gas with out liquid state is called sublimation. Similarly conversion of gas into solid without liquid state is also called sublimation. The product formed as a result of sublimation is called sublimate. The substances that undergo sublimation are camphor, Ammonium chloride, Naphthalene etc.
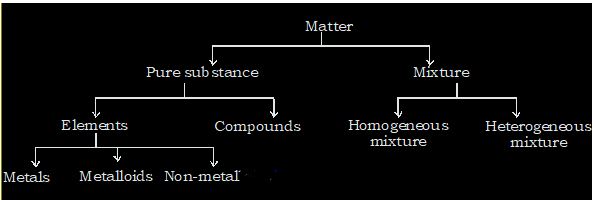
Classification of matter on the basis of chemical composition:
On the basis of chemical behaviour, matter can be classified into pure substance and mixture. A homogeneous material which contains particles of only one kind and has definite set of properties is called a pure substance.
In case of pure substance, the molecule may contain similar or dissimilar atoms. Molecules which contain atoms of same kind are called elements and molecules which contains atoms of different kind are called compound.
If two or more substances are mixed together in any proportion and if they retain their characteristics, the resulting mass is called mixture.