-
Interconversion Of Matter
SOLVED EXAMPLES
Question:Convert the following temperature to Celsius scale :
(a) 300 K (b) 573 K
Solution:We know that 0C = K – 273
(a) 300 K = 300 – 273 = 270C
(b) 573 K = 573 – 273 = 3000CQuestion:What is the physical state of water at :
(a) 250C (b) 00C (c) 1000C ?
Solution:(a) At 250C, physical state of water is a liquid.
(b) At 00C, physical state of water can be either a solid (ice) or a liquid.
(c) At 1000C, boiling point of water, water exists both as a liquid as well as a gas.Question:For any substance, why does the temperature remain constant during the change of state?
Solution:The temperature remains constant during the change of state of a substance at its melting point or boiling point because, heat energy supplied to the substance is used up in overcoming the forces of attraction between the molecules. As a result, the temperature does not rise till the entire substance undergoes change of state.Question:Suggest a method to liquify the atmospheric gases.
Solution:Atmospheric gases can be liquified either by increasing the pressure or decreasing the temperature (i.e. by cooling). This is because, by doing so, the forces of attraction between gas particles become so high that they bind these particles together to form the liquid state.Question:The Naphthalene balls disappear with time without leaving any solid. Give reason.
Solution:Naphthalene undergoes sublimation slowly at room temperature. As a result, solid naphthalene gets converted into vapours which become a part of air around us. Therefore, naphthalene balls disappear without leaving any solid.Question 18: Why is ice at 273K more effective in cooling than water at the same temperature?
Solution: At 273K, ice can absorbs more amount of heat than water due to its latent heat of fusion. In other words, the ice at 273K has less heat energy than water at same temperature and hence ice is more effective in cooling than water at the same temperature.Question 19: What produces more severe burns, boiling water or steam ?
Solution: The steam has more heat energy stored in it than in boiling water at the same temperature (i.e at 373K) due to latent heat of vaporization. Hence, steam will produce more severe burns than boiling water.Question 20: Name A, B, C, D, E and F in the following diagram showing change in its state :
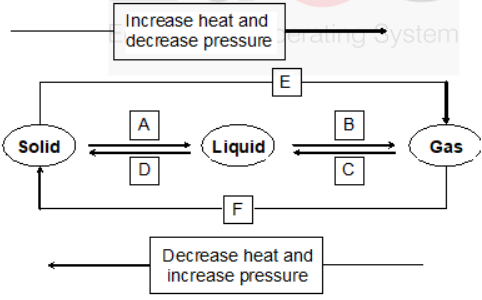
Solution: A – Fusion
B – Vaporisation
C – Condensation
D – Solidification
E – Sublimation
F – Sublimation -
Interconversion Of Matter
SOLVED EXAMPLES
Question: Why does a desert cooler cool better on a hot dry day ?
Solution: A hot dry day means temperature of the atmosphere is high and humidity of air is low. Since both these factors increase the rate of evaporation, an enormous cooling is produced and thus desert cooler cool better on a hot dry day.Question: How does the water kept in an earthen pot (matka) become cool during summer?
Solution: During summer, water kept in an earthen pot (matka) continues to evaporate through the fine pores on it. Since evaporation causes cooling, water kept in matka becomes cool during summer.Question: Why are we able to sip hot tea or milk faster from a saucer rather than a cup ?
Solution: The surface area of the liquid hot tea or milk is more in a saucer than in a cup. Therefore, evaporation or cooling will take place more rapidly in a saucer than in a cup. Consequently we are able to sip hot tea or milk faster from a saucer rather than a cup.Question: What type of clothes should we wear in summer?
Solution: In summer, we sweat or perspire more. The cotton clothes, being good absorber of water, absorbs the sweat and exposes it to the atmosphere for easy evaporation. Since evaporation causes cooling, therefore, we should wear cotton clothes in summer.