-
Chemical Reactions
CHEMICAL DISPALCEMENT REACTIONS
A single-displacement reaction, is a chemical reaction in which one (or more) element(s) replaces an/other element(s) in a compound. It can be represented generically as
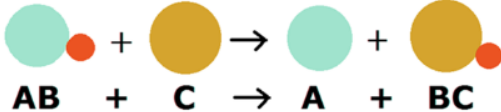
This will most often occur if A is less reactive than C, thus giving a more stable product.
All simple metal with acid reactions are single displacement reactions. For example, the reaction between magnesium, Mg, and hydrochloric acid, HCl, forms magnesium chloride, MgCl2, and hydrogen, H2.
\( Mg_{(s)} + 2HCl_{(aq)} \to H_{2(g)} + MgCl_{2(aq)} \)
Reactivity order of metals:
LI<K<Na<Ca<Mg<Al<Zn<Fe<Pb<Sn<[H]< Cu<Hg<Ag<Au<Pt.
Fe+CuSO4 (aq) \( \to \) FeSO4 (aq) +Cu
Here Fe is more reactive than Copper so it displaces copper from copper sulphate solution.EXAMPLE
When chlorine is added in its gaseous form (or as a gas dissolved in water) to the solution of sodium bromide,solution turns brown.This is because,the chlorine displaces the place of bromine and therefore,we can say chlorine is more reactive than bromine.The brown colour of the solution is due to the displacement of bromine.
The equation can be written as follows
Cl2(aq) + 2NaBr(aq) \( \to \) 2NaCl(aq) + Br2(aq)
chlorine + sodium bromide \( \to \) sodium chloride + bromine
A chemical displacement reaction can be better understood by another example
Dissolve a little amount (0.5 gm) of Silver nitrate in 10 ml of water in a test tube. A copper wire is then dipped in it and kept undisturbed for some time. Now, we can observe shining silver crystals which are visible on the Copper wire.This is because the copper metal displaces silver from Silver Nitrate solution.Also,the solution becomes bluish as some amount of copper is developed.The equation can be written as follows:
Cu(s) + 2AgNo3(aq) \( \to \) 2Ag(s) + Cu(NO3)2(aq)
copper + silver nitrate \( \to \) silver + copper nitrate